 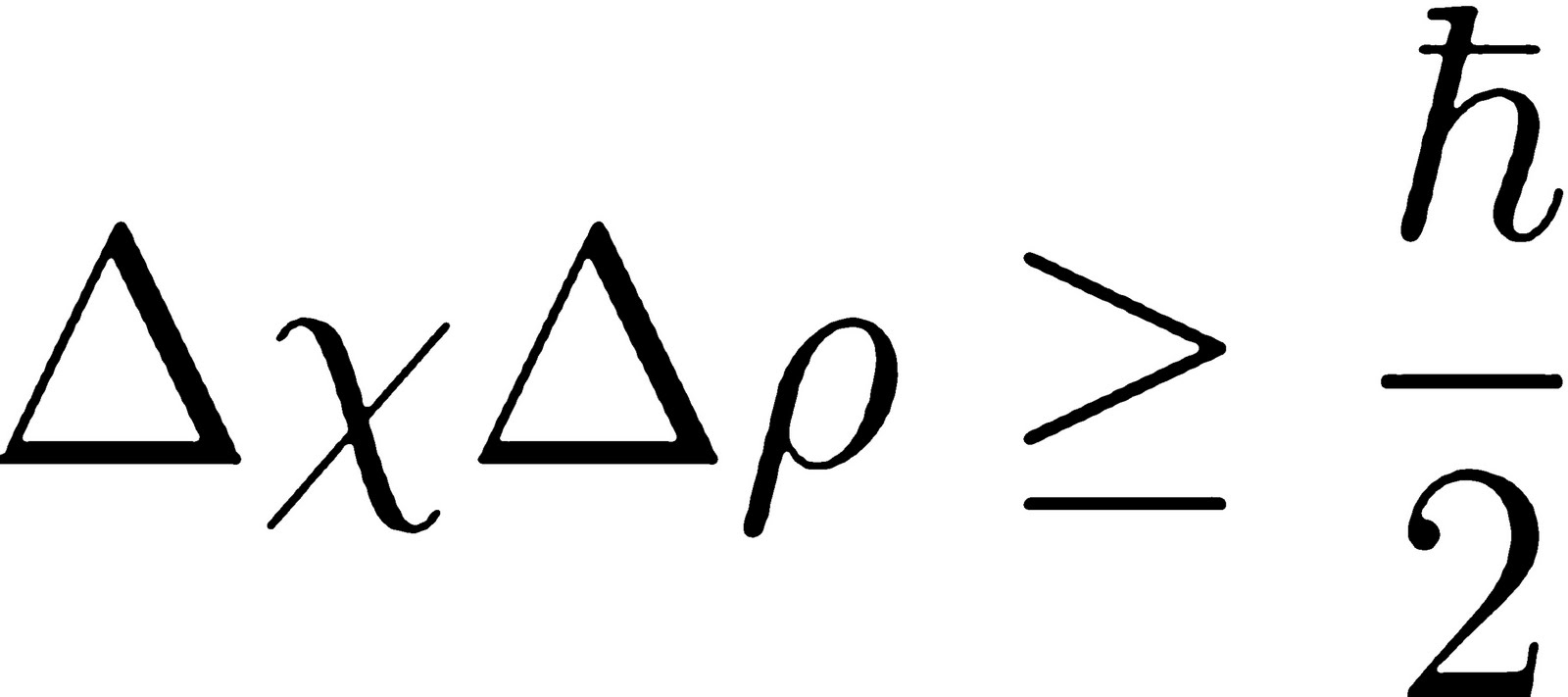
You don't know the momentum, or you don't know the If you know where that particle is in space really well, Uncertainty principle, you can't know the position and momentum of that particle accurately,Īt the same time. Particle, the linear momentum is equal to the Mass times the Velocity. M, moving with Velocity V, the momentum of that Particle, let's say we have a particle here of Mass Heisenberg uncertainty principle is a principle of quantum mechanics. The rule is that the product of the uncertainties of the position and momentum cannot be less than h/(4π) = 5.27286×10⁻³⁵ J∙s (h is the Planck constant). So, it is not MERELY a matter of our not being able to measure with arbitrary precision, but that the particle itself does not HAVE an exact momentum nor an exact location within very specific rules. Likewise, the more set the position becomes the less set the momentum of the particle actually is. Completely apart from any measurement anyone might make, the more set the momentum becomes the less the particle even HAS a set location. 
This is a limit imposed by nature and it can never be overcome as far as we know with current understanding of nature. Thus, while we can refine instruments to some extent, it is utterly impossible to construct any instrument that would overcome this problem. Likewise, any mechanism by which we might measure the electron's position will affect its momentum, making the momentum less certain. It isn't so much that we cannot measure both momentum (and it is momentum, not velocity) and position of an electron at the same time, but rather there is a limit placed by nature on how precisely we may simultaneously measure both.Īny mechanism by which we might measure the electron's momentum will affect the position, making the position less certain. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
